
Surge Immunity Testing LAB
Surge immunity testing is a test project under EMS, which mainly evaluates the immunity of the EUT (Equipment Under Test) to transient induced voltages caused by lightning.
During the operation of the EUT, lightning strikes can induce high peak voltages on the power and signal lines. These voltages can cause a performance degradation or even damage the EUT. Surge testing simulates this scenario by using a surge signal generator to produce a spike voltage, which is coupled onto the power and data lines. The test observes whether the EUT can operate normally under such a high induced voltage impact.
JJR Laboratory in China is a third-party emc testing agency. The laboratory has testing qualifications and capabilities for GB/T 17626, iec 61000-4-5, en 61000-4-5, GB/T 18595, CISPR 14-2, EN 55014-2 standards, and can provide third-party lightning surge immunity testing services.
Surge Immunity Testing Overview
Surge immunity testing is used to simulate the effects of pulses generated by natural lightning or when large capacitive loads are introduced into the power grid. It includes both power and signal line tests.
Power Line Testing
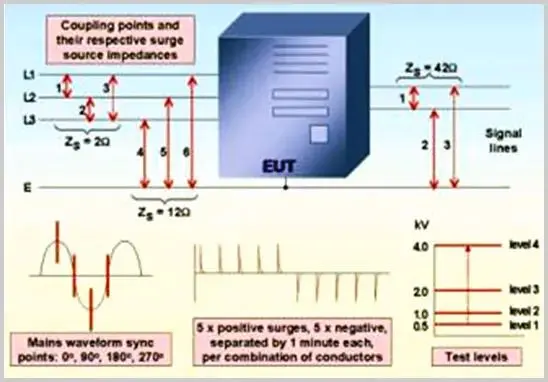
Includes tests between L and N lines, L to protective earth, N line to protective earth, and L&N to protective earth. The first case is differential mode interference, while the latter three are common mode interferences.
Signal Line Testing
For shielded cables, the interference is applied to the shield layer. For ground wires, the interference is applied to the signal line. For user lines, the interference is applied directly to the A and B lines.
Surge Immunity Testing Standards
- GB/T 17626.5-2008
- IEC 61000-4-5:2014+A1:2017
- EN 61000-4-5:2014 “Electromagnetic Compatibility - Testing and Measurement Techniques - Surge (Immunity) Tests”
- CISPR 14-2:2015
- EN 55014-2:2015 "Electromagnetic Compatibility Requirements for Household Appliances, Electric Tools, and Similar Appliances - Part 2: Immunity"
- GB/T 18595-2014 “Electromagnetic Compatibility Immunity for General Lighting Equipment”
Surge Immunity Testing Equipment
- Test Generator
- Coupling Devices
- Decoupling Networks
- Ground Reference Plane, etc.
Testing Environment
- Temperature: 15°C ~ 35°C
- Relative Humidity: 10% ~ 75% RH
- Atmospheric Pressure: 86 kPa ~ 106 kPa
Test Waveforms
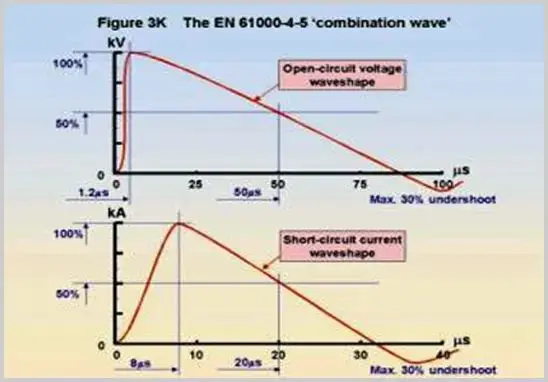
Surge waveforms include: 1.2/50(8/20) combination wave and 10/700 voltage wave. The 1.2/50 and 1.2/50(8/20) waveforms are used for power line and indoor signal line testing, while the 10/700 voltage wave is used for outdoor signal line surge tests.
In the above waveforms, 1.2, 8, and 10 refer to the rise time (in microseconds), and 50, 20, and 700 refer to the pulse width (in microseconds). The energy in surge waveforms is much higher than that in EFT/B and ESD, but the interference bandwidth is much narrower.
Why Choose Guangdong China JJR Laboratory for Testing?
1. Trusted Brand: Established in 1998, a high-tech enterprise accredited by China CNAS and CMA, and a national public technical service platform for SMEs.
2. Multiple Certifications: Recognized by multiple domestic and international agencies, with comprehensive qualifications and internationally credible test reports.
3. International Standards: The laboratory operates according to international standards ISO/IEC17025 and complies with more than 10 international and domestic standards.
4. Service Assurance: Efficient one-stop testing and certification services, with complete test plans, trusted by numerous well-known domestic and international companies.
Email:hello@jjrlab.com
Write your message here and send it to us
 What Are the Safety Tests for Lithium Batteries?
What Are the Safety Tests for Lithium Batteries?
 Wireless Device EMC Certification
Wireless Device EMC Certification
 Type-C Interface EN62680-1 Testing
Type-C Interface EN62680-1 Testing
 Electrical Safety Test IEC 60601
Electrical Safety Test IEC 60601
 IEC 60601-1-2 EMC Testing
IEC 60601-1-2 EMC Testing
 Polycyclic Aromatic Hydrocarbons (PAHs) Testing La
Polycyclic Aromatic Hydrocarbons (PAHs) Testing La
 EU EN71 Certification Testing for Mechanical Toys
EU EN71 Certification Testing for Mechanical Toys
 EU Toy Safety Testing (EN 71 / 2009/48/EC)
EU Toy Safety Testing (EN 71 / 2009/48/EC)
Leave us a message
24-hour online customer service at any time to respond, so that you worry!




